About us
Creating value for customers with professional application technology and technical support
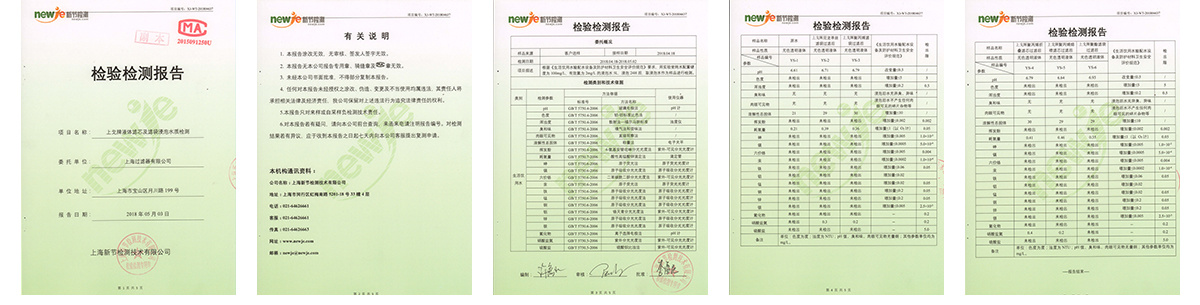
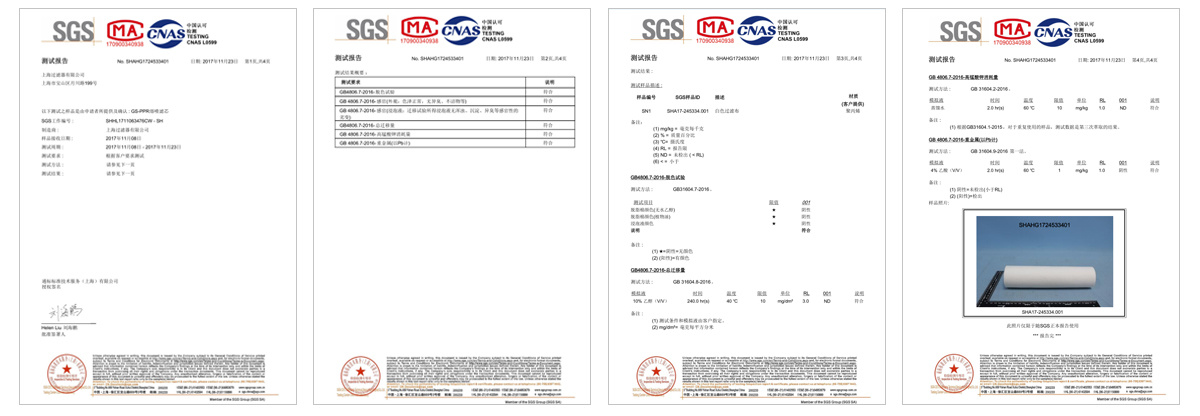
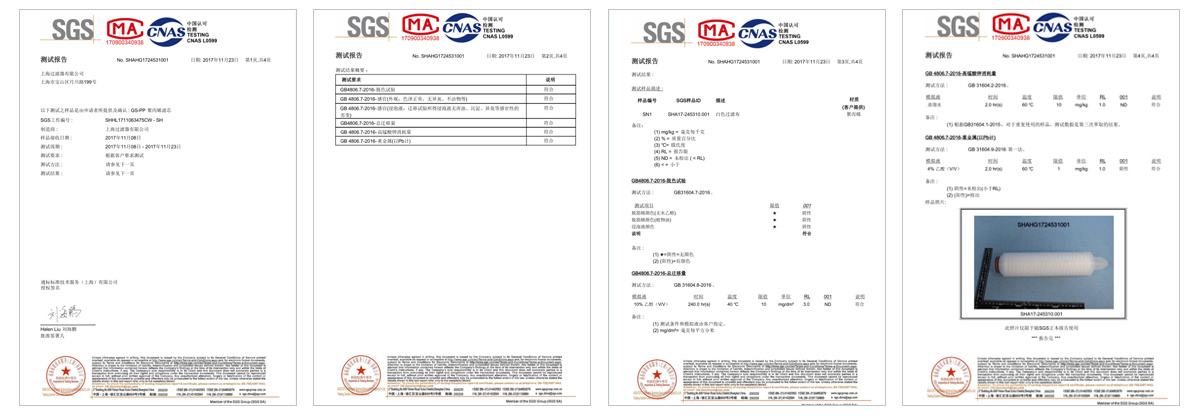
Testing
Shanghai Filter co., LTD. USES the international advanced experimental instrument, according to the related standard and the regulation and the strict management procedure establishment laboratory, devotes to the new product development and the development as well as the quality control continuous improvement, has guaranteed the product the reliability and the stability greatly. At the same time, the company's verification laboratory, in cooperation with domestic authoritative third-party verification companies, can provide users with neutral and professional filter core performance process verification services.
|
Standards to which the cartridges conform and regulations followed.
-GB 9688-88, GB/T 5009.60-2003 and GB/T 17219-1998
-GB/T 5750-2006 and GB/T 14233.1-1998 -Meets USP 121°C Class VI plastic test -No fibre shedding, in accordance with FDA 21 CRF regulations -Bacteria removal cartridge passed ASTM F-838-05 liquid bacteria challenge test -Water-related hygiene permit for cartridge -Cartridge tested by Shanghai Centre for Disease Control and Prevention -Cartridge passed the SGS EU ROHS 2.0 test -Cartridge passed SGS EU Total Migration Test for food-grade water phase |
 |


Information
Tel:+86 21-56151124
Fax:+86 21-56807944
E-mail:zzy@sh-filter.com.cn
Add:NO. 515,Cenglin Road,Lin-gang Special Area,China (Shanghai) Pilot Free Trade Zone
Fax:+86 21-56807944
E-mail:zzy@sh-filter.com.cn
Add:NO. 515,Cenglin Road,Lin-gang Special Area,China (Shanghai) Pilot Free Trade Zone
Powered by 300.cn The site is IPV6 enabled


